Researchers discover how morphine relieves pain in the brain and create a gene therapy that mimics its effects without addiction risks. A breakthrough that could transform chronic pain treatment.
You hit your toe, and right away it feels like the worst pain ever. But the strange thing is, the pain isn’t exactly the same as the actual injury. Scientists say pain works in a weird way, it’s not just about what your body feels. There’s also an emotional part to it, like a sudden fear or discomfort that makes you want to be extra careful with your toe afterward.
For people who have chronic pain, like from nerve damage, arthritis, or after surgery, the emotional side of pain can become overwhelming. It’s not just that their body hurts all the time. Their brain starts treating the pain like a constant danger, almost like an alarm that won’t turn off because of which, they may stop doing things they enjoy and pull away from everyday life.
Drugs like morphine only reduce the physical pain, but also calm that alarm feeling in the brain. So the pain might still be there, but it doesn’t feel as scary or important.
The problem is, these drugs can be risky. Over time, people may need more of the drug for the same effect (this is called tolerance). They can also become dependent on it or even addicted. In serious cases, taking too much can slow breathing and lead to overdose.
A new study published in Nature offers a solution, a way to replicate morphine’s emotional pain relief without its dangers. And it all comes down to a tiny cluster of neurons hidden deep in your brain’s pain center.
The Brain’s Pain Headquarters
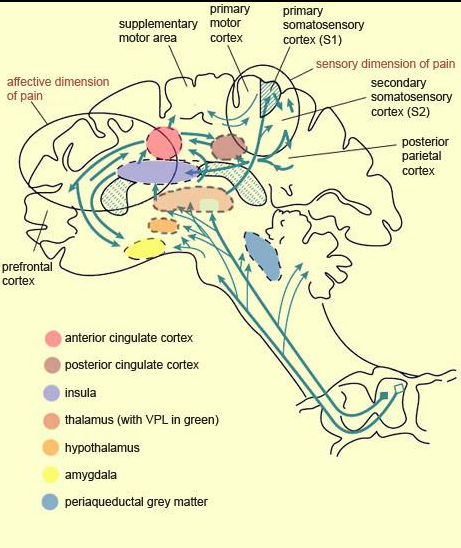
Buried beneath your forehead, tucked into the folds of your frontal lobe, sits a region called the anterior cingulate cortex (ACC). This is where your brain decides whether something matters. It’s involved in emotional processing, decision-making, and also the unpleasantness of pain.
When you’re injured, raw signals race up your spinal cord about tissue damage. But before those signals even reach your conscious mind, your ACC is already deciding:
“This is bad. I need to do something about it”.
This emotional interpretation is what makes pain painful. It’s what makes you want to pull your hand away from a hot stove not just automatically, but because you’re afraid of getting burned.
Neuroscientists have suspected for years that opioid drugs might work here, at this emotional level, rather than just numbing sensation. But proving it required a level of precision that hasn’t been possible until now.
How Researchers Peered Inside the Pain Machine
The researchers, led by teams at the University of Pennsylvania, Stanford, and other institutions, combined deep learning AI with direct neural recordings in mice experiencing pain.
First, they needed to observe how animals act when they’re in pain but there was a problem. The usual way scientists test pain in animals is pretty simple, they might touch them with something and see how quickly they pull away.
That kind of test only shows a basic reaction, like a reflex. It doesn’t really tell us how the animal feels or whether the pain is actually bothering it in a deeper way. The emotional side of pain, the part that makes it stressful or hard to live with, is missing from these kinds of tests.
So the team invented LUPE (Light Automated Pain Evaluator), a dark chamber where mice could move freely while high-speed cameras tracked their every movement using artificial intelligence. Not just whether they moved, but how they moved, when they moved, and what that pattern of movement reveals about their internal state. Gregory Corder, the lead researcher said,
“Pain isn’t just about the reflexive response. It’s about motivation. It’s about whether an animal wants to engage with life or withdraw”.
The AI system decoded six basic behaviors (stillness, walking, rearing, grooming, and paw-licking) and then used statistical analysis to identify six distinct “pain states”, hidden emotional and motivational conditions that weren’t obvious from any single behavior. It’s like decoding a language made of movement.
When they gave morphine, they observed that the drug didn’t affect all pain states equally. Instead, it specifically suppressed one particular state, one that captured the animal’s affective-motivational response to pain. The sensory part was left untouched and the reflexive pulling away was unchanged. Just the suffering, dialed down.
The Neurons That Morphine Hypnotizes
Armed with this behavioral insight, the researchers then turned to the microscopic level. They looked inside the ACC pain region and marked which neurons were active during pain. Then they sequenced the genetics of those neurons to see what made them special.
They concluded that about 70% of these pain-encoding neurons expressed a μ-opioid receptor, the molecular lock that morphine’s key fits into.
Even more remarkably, in chronic pain states (induced by nerve injury in the mice), these specific neurons remained morphine-sensitive even as the brain underwent other chronic pain adaptations.
Eventually, when researchers genetically deleted these opioid receptors specifically from the ACC in one group of mice, morphine stopped working. The drug no longer reduced the affective-motivational pain response. But when they reintroduced opioid receptors back into the ACC of mice that lacked them entirely, morphine’s pain-relieving effects came roaring back. Corinna Oswell one of the researcher conducting the study said,
“We found the specific neurons, the specific circuit. This is where morphine does its emotional pain relief work”.
From Discovery to a Safer Drug
Once you understand how something works, the next step is usually asking, can we do it better?
Morphine and other opioid drugs are broadcast signals. You take a pill, and these opioid molecules flood your entire nervous system, binding to opioid receptors everywhere,
- in your gut (causing constipation)
- in your respiratory system (potentially stopping breathing)
- in reward centers (leading to addiction)
It’s a sledgehammer when what we really need is a scalpel.
The research team designed a scalpel.
They created a piece of genetic code that targets only opioid-receptor-expressing neurons in the ACC. They packaged this into a virus and used it to infect those cells with a new molecular tool, an inhibitory protein that can be turned on and off with a drug called deschloroclozapine (DCZ).
The specificity is almost impossible to overstate. The viral vector only infects cells expressing opioid receptors. The genetic promoter only allows the inhibitory protein to be expressed in cells with those receptors. When you give DCZ, it selectively silences only these opioid-sensitive pain neurons.
In chronic pain models (nerve injury), this chemogenetic therapy worked. Mice that received DCZ showed reduced affective-motivational pain responses. They licked their injured paws less. They spent less time in pain-associated behavioral states. The relief lasted without tolerance developing, animals didn’t need higher and higher doses as their bodies adapted.
Most strikingly, when researchers tested side effects using place preference tests (essentially asking, does the animal find this experience rewarding?), the therapy didn’t trigger addiction-like behavior in healthy animals. In injured animals, the relief was reinforcing, they sought it out, but that’s because pain was relief, not because the treatment itself was drugs being rewarding.
What This Tells Us About Pain Itself
Beyond the therapeutic potential, this work reveals something profound about how your brain creates the experience of pain.
Pain isn’t one thing. It’s at least two:
- the sensory dimension (knowing tissue is damaged)
- the affective-motivational dimension (feeling that damage is a problem you need to address).
For acute pain, this dual system is brilliant, it both informs you of danger and motivates protective action.
But in chronic pain, this system malfunctions. The brain learns to amplify the threat. Pain neurons become hyperexcitable. Emotional pain centers become stuck in a heightened state. The result is suffering that persists long after healing should be complete.
What morphine reveals, and what this gene therapy confirms, is that we can modulate that emotional layer without destroying the sensory one. You can still know your leg is injured, but you don’t have to dread it. You can still pull away from extreme heat, but you don’t have to be consumed by the fear of heat. That distinction, that we can modulate affective-motivational pain while leaving sensation intact, opens up entirely new ways of thinking about pain treatment.
When AI Meets Neuroscience
One element of this research that shouldn’t be overlooked is how it was done. The LUPE behavioral analysis system represents a shift in how neuroscience can work. Traditional animal studies were limited by observer bias and the sheer labor of hand-scoring behavior. One researcher could manually score one mouse’s behavior in 50-150 minutes. The same work in LUPE takes seconds, across dozens of animals simultaneously.
This makes possible the kind of large-scale, longitudinal studies that reveal how the brain actually works in semi-natural conditions. It allows researchers to ask better questions and get clearer answers. And it demonstrates that the future of neuroscience isn’t just better microscopes, it’s also smarter software.
The Takeaway
The path from this discovery to a patient receiving treatment remains long and uncertain. Gene therapies require regulatory approval and safety validation along with manufacturing scale-up. Long-term effects in human brains need careful study.
But the conceptual problem has been solved. Scientists now understand the specific circuit in the brain that makes pain unpleasant, and they’ve proven they can target it with remarkable specificity. They’ve shown that this approach can mimic the emotional pain relief of morphine while avoiding the worst side effects.
The next act of this story will be written in human clinical trials but for the first time, we have a map showing where to go.
Publication details:
Oswell, C.S., Rogers, S.A., James, J.G. et al. Mimicking opioid analgesia in cortical pain circuits. Nature 649, 938–947 (2026). https://doi.org/10.1038/s41586-025-09908-w
Frequently Asked Questions
Q: How is pain sensation different from emotional suffering?
Pain has two parts: the sensory signal (your finger touches a hot stove) and the emotional response (the dread that makes you pull away). This research targets only the emotional part, leaving protective sensation intact. It’s like still knowing the stove is hot, but not being consumed by fear of it.
Q: If I use this therapy, will I still feel pain?
Yes, you’ll still feel and react to harmful stimuli. The therapy reduces the emotional dread of pain, not the sensation itself. You’ll still pull your hand from fire, you just won’t obsess over the injury afterward.
Q: How does morphine currently work in the brain?
Morphine binds to opioid receptors in pain-emotion circuits (especially the anterior cingulate cortex), dampening the brain’s threat alarm. The problem is morphine’s effects spread throughout the body, causing breathing problems, constipation, and addiction risk. The new gene therapy targets only specific pain neurons.
Q: Why do people need higher opioid doses over time?
Your nervous system adapts to continuous opioid presence, a process called tolerance. Your neurons reduce opioid receptors or make downstream signaling less responsive, similar to how ears adapt to loud music. The new gene therapy approach may avoid tolerance because it creates signals from inside neurons, not from external drug molecules.
Q: What is the anterior cingulate cortex and why is it important?
The ACC is a small brain region involved in emotions and decision-making. It’s where your brain decides whether pain is a crisis or just a sensation. Brain imaging shows ACC activity directly predicts how much pain bothers you emotionally, making it the ideal target for precision pain relief.
Q: How does this differ from current pain treatments?
Traditional opioids flood the entire body, causing widespread side effects and addiction risk. This gene therapy uses a virus to deliver a genetic “off switch” to only opioid-sensitive neurons in the pain center. It’s circuit-specific, cell-type-specific, can be controlled on-demand, and shows no tolerance in early studies.
Q: When will humans be able to get this treatment?
Not for at least 5-10 years. The research is at proof-of-concept, human trials haven’t started yet. Researchers still need to solve delivery challenges (currently requires brain injection in mice) and conduct safety studies. But unlike many basic research, this has a clear path forward through FDA approval.
Q: What are the risks or side effects?
Early studies show high specificity (97% of targeted cells), no tolerance, and no addiction-like behavior in healthy animals. Potential concerns include immune response to the viral vector and uncertainty about long-term effects (gene therapies are permanent). Ongoing research will clarify risks before human trials begin.
Q: Could this approach treat other conditions like depression or anxiety?
Possibly, the same circuit-targeting approach could theoretically address other emotional conditions. But each would need separate research. This work is laser-focused on pain because the neural circuits are best-understood there.