The idea that nanobots flowing through our blood streams to deliver localised medication or to detect any tumour formation is no more a far-fetched dream. A group of researchers from the Max Planck Institute for Intelligent Systems (MPI-IS) in collaboration with the Max Planck Institute for Solid State Research (MPI-FKF), have developed microswimmers that can navigate through biological fluids, areas that are otherwise difficult to access. Steering is done through external form of light energy.
Even in case of salty and highly acidic environment, the microswimmers derived from organic microparticles, have shown promising results. Designing such type of advanced microswimmers was quite challenging.
Salt content present in the bodily fluids usually obstructs the light energy powered locomotion in the nanobots. Therefore, to scale up the concept is not an easy task. Also, manoeuvring the nanobots from outside the body is not only challenging but it also cost a lot of money.
To make it a programmable undertaking, researchers employed porous two-dimensional organic carbon nitride (CNx). The polymer is extracted from urea. Carbon nitride can absorb light – like the solar cells of photovoltaic panel – that helps the bots in propulsion. When light touches the bot’s surface, the tiny device moves forward.
High ion tolerance
According Filip Podjaski, from the Nanochemistry Department at MPI-FKF, salt concentration in biological fluids prohibits the bot’s movement so to counteract the problem, researchers exploited the porous structure of the nanoparticles (CNx). The spatial arrangement of the particle helps free flow of ions through it, thus, lessening the resistance that is otherwise offered by salt components.
Additionally, external light allows mobility of ions in CNx microswimmers, consequently, the particle moves with great agility.
After making the CNx microswimmers salt tolerant, researchers moved to the next level that is, used them as drug carriers. They burdened the pores with an anti-cancer drug Doxorubicin. Scientists observed that the microbots were able to sustain the drug load for more than a month. Following which, they were able to release drug in fluid that was rich with acidic pH level.
Accumulating the light energy intrinsically
While they were experimenting, researchers observed that when the drug cargo encountered acidic conditions, as in when it is in stomach – where by default the pH level is acidic – immediately, the drug is released in high amounts.
Podjaski added that the exposure of blue light not only aids in propulsion but it also triggers the release of the carried drug. Therefore, to solve this problem, the intrinsic charging ability of the carbon nitride comes into action.
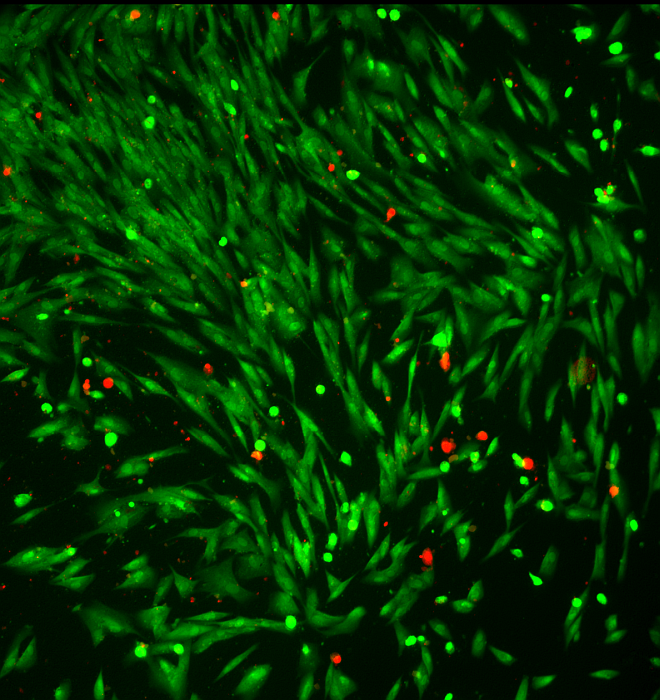
The material charges up when light is exposed to it in oxygen depleted (hypoxic) environments. Like any regular solar battery, the material can accumulate the light energy intrinsically. Therefore, capacity of the material to charge in hypoxic conditions, also triggers it for the drug release.
Real tumor cells were used to conduct the study.
Takeaway
As per Metin Sitti from the team said the experiment opened up doors for unanticipated potential from easily synthesizable, abundant and porous microparticle materials. Previously it was used in applications such as photocatalysis.
The porosity has helped the researchers in carrying heavy drug load without using or fabricating any new encapsulation structures, which otherwise is also difficult to control. Microswimmers developed from biocompatible materials are a vision of the future and application is unlimited from inner biological structures to interplanetary probes and intelligent semi-autonomous systems, micro bots could be used everywhere.