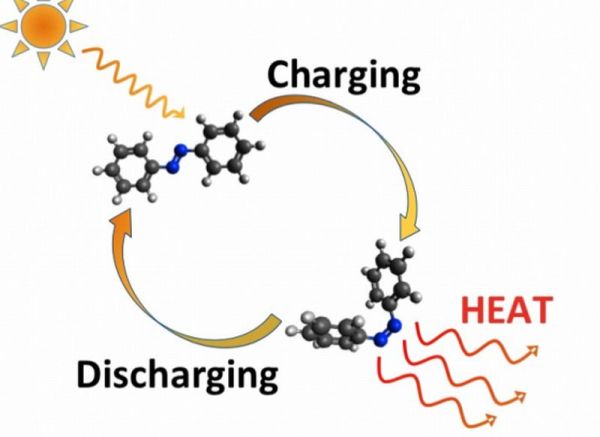
Researchers from MIT and Harvard University have come up with an innovative approach of tapping sun’s heat in form of chemical energy and releasing as per the requirement.
The technique is not aimed for replacing the existing solar energy storage problem rather it is an effort of taking the solar power into the new areas which otherwise was not explored before. For instance, the output via this approach is heat, which could be directed towards cooking or industrial avenues where heat is required for powering processes.
Molecules Act as Rechargeable Thermal Batteries
Azobenzene, the photoswitching molecules that are the building blocks for this technology plays dual role in the entire process. Upon receiving sunlight, they absorb energy and jump from their initial configuration to a new level. There they can stay at the state of equilibrium for relatively longer time span. However, upon experiencing fraction of heat, light or even electricity, they reiterate to their initial phase of course giving out heat.
The principle is quite similar to rechargeable thermal batteries, where they get excited upon receiving solar energy, goes to the next level storing that energy and then giving away the accumulated energy as and when required.
The Approach an Extension of Grossman’s Work
This approach is not a new one, Grossman the Carl Richard Soderberg Associate Professor of Power Engineering at MIT and his team started the theoretical work three years ago. Going to the practical level was a little bit tricky since the exact amount of energy density that could be stored in a given material required precision for an efficient carrying out of the experiment. Secondo, the arrangement at molecular level was too squeezed up, again that was not as easy as it looked via computer simulations.
During the preliminary phase, Grossman’s team struggled with fixing the molecules to carbon nanotubes (CNTs), which was too difficult to yield. However, they were able to reach a point where the packing density was less than half of what the computer simulations predicted yet the material was releasing enough heat storage that they were actually looking for. This came as a surprise to them since, the practical level workings demonstrated the desired amount of heat discharge even with lesser amount of energy density, this instigated them for further research.
Interleaving Azobenzene Resulted in Larger Enhancements
While fixing the photoswitching molecules or the azobenzene the researchers discovered that some of the molecules were protruding from the sides of CNTs but like comb, the individual teeth were wide apart. So to take the advantage of the space within the molecules, the team interleaved the azobenzene molecules attached to adjacent CNTs consequent upon which, the squeezing up of the molecules resulted automatically and with lesser effort.
This idea of sandwiching the azobenzene molecules between adjacent CNTs proved meticulously well and from initial 30 percent the proportion of energy storage escalated to 200 percent.
Revolutionizing Solar Thermal Materials
Researchers claimed that this experiment has open up a new class of solar thermal materials that would take the solar energy in completely new paradigm. Areas where electricity is not required, this material would take the role of generating heat, it will work wonders in arenas like powering heat-based industrial processes and cooking fuel.
Generally, fuels are burnt while performing action but in this case, there would be no emissions and since it will be reused so nothing will be consumed technically.
The system is still not ready for practical applications yet, however, research is still being done to make it a viable system.