Human body is composed of different biological cells. Each cell is unique. Research shows that a sample of biopsied tumor consists of various distinct cells. These cells differ in their rate of proliferation, responsiveness to drugs, potential metastasis and so on. Therefore, it is essential for researchers to understand the behavior of individual cells and then accordingly design therapy and treatments, for which seizing individual cell is very important.
Tools that are available now to capture individual cells of interest are not very accurate and data pooled from a tissue samples are averaged over a swarm of cells. However, researchers from the US Army Research Laboratory in Adelphi and The Johns Hopkins University in Baltimore have finally created a small sized self-folding gripper. This gripper is capable of seizing individual cells under in vitro, as well as in vivo conditions.
The gripper can be produced in mass like 100 million present on a 12-inch wafer, which can be sent to a specific body part to seize certain types of cells. These gripper works similar to Venus fly-trap that capture its prey, by slowly closing its arm around interest cells, but without harming them. During a trial, researchers using the grippers could trap mouse fibroblast cells and red blood cells under in vitro condition.
The newly designed gripper is very effective in capturing single cells of interest, unlike the traditional single cell analysis techniques as optical, microfluidic trap, microwells, flow cytometry and certain tiny robotic devices that often lose their grip and additionally require wires and tethers that limit their movements and use.
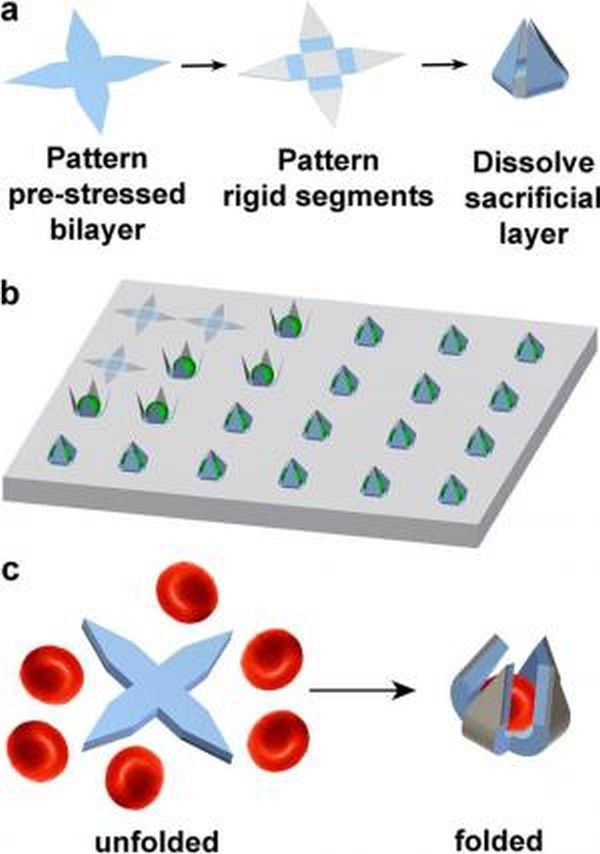
The self-folding gripper is capable of capture cells using energy that is released due to stress in its own material, eliminating the requirement of wires, tethers or batteries. These grippers are efficient as the gripper’s hinges are composed of bilayer of pre stressed SiO/SiO2. The hinges are attached to a body and arms composed of only SiO. These SiO and SiO2 are biocompatible and bioresorbable and over the time get dissolved in the body’s biological fluids. When introduced to a saline solution, the atoning layer releases the gripper arms, which curl upward and enclosing a cell.
Researchers using photolithography showed that the self folding grippers can be created in sizes measuring between 10 to 70 µm from tip to tip when open. This size is optimal to seize various types of individual cells. Grippers could get folded at angles starting from 90 degrees to 115 degrees by manipulating the thickness of bilayer film. Moreover, the opening at the arm’s intersection helps in the free flow of nutrients, waste and biochemical, in and out of the cells, without killing the cells. And as the grippers are optically transparent, they can be placed under optical microscope to study the trapped cells. But researchers have not yet able to control the timing of gripper to seize specific chemical or cells.
Researchers are working to control and time the gripper to capture interest cells. Once this is achieved, these tiny grippers can be sent to different and hard to reach body parts to collect cells. For uses under in vivo conditions, grippers can be directed using ferromagnetic elements and biomarkers to target diseased cells. Under in vitro condition, magnetic fields as nickle can be used to direct grippers. Researchers suggest that in the future, tiny grippers have an important role to play in different areas of medical science. Understanding how disease cells behave, researchers can design therapies for targeted cells.